اتم چیست؟ سوالی در مورد فیزیک! مدیاسافت
Calcium Bohr diagram Calcium
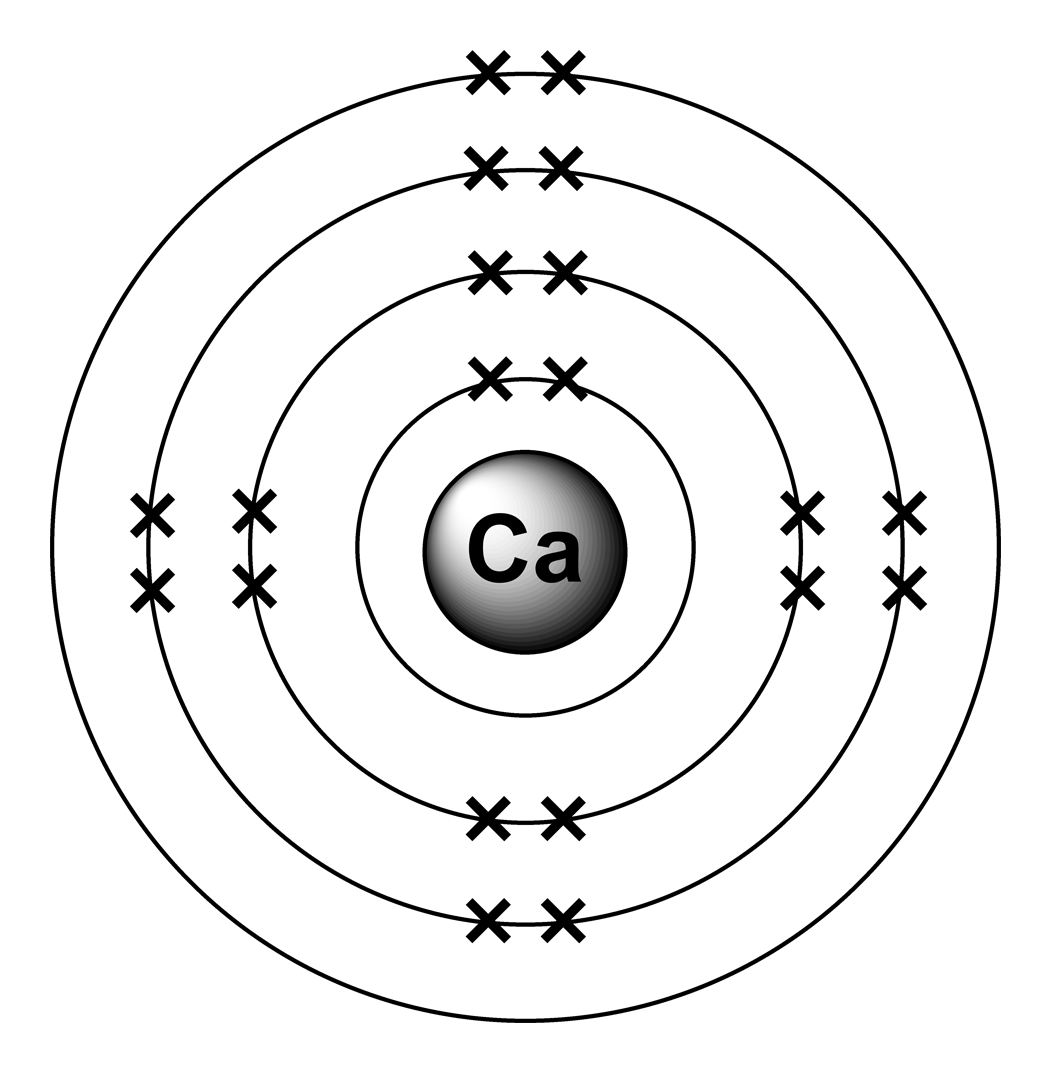
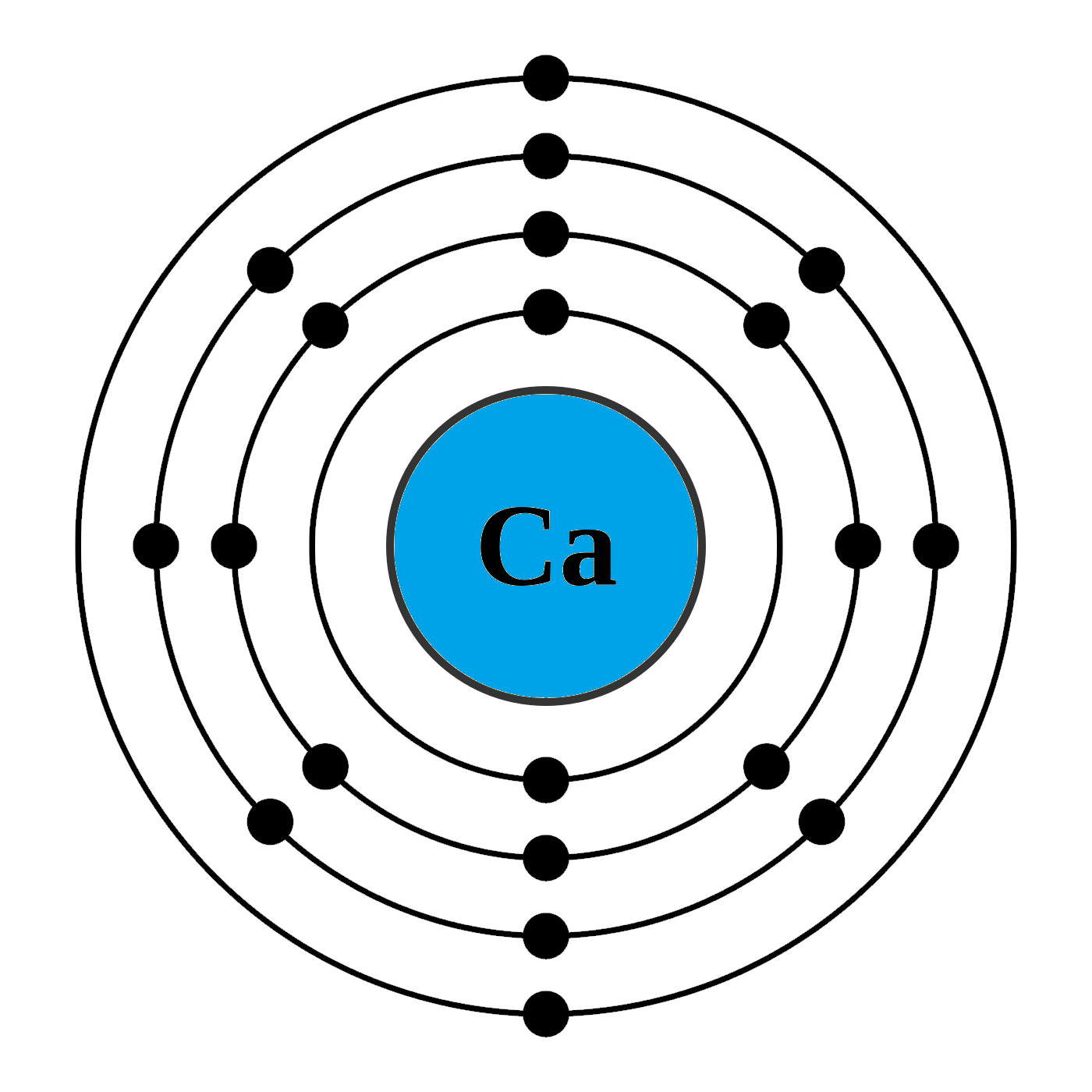
Calcium has 2 electrons in its first shell, 8 in its second, 8 in its third, and 2 in its fourth.Check me out: http://www.chemistnate.com
Bohr Diagram Of Calcium
The Bohr model of the hydrogen atom explains the connection between the quantization of photons and the quantized emission from atoms. Bohr described the hydrogen atom in terms of an electron moving in a circular orbit about a nucleus.. Suggest a reason for the observation that the spectrum of calcium is more complicated than the spectrum of.

27 Bohr Diagram For Calcium Wiring Database 2020
Why is the valency of the Calcium (Ca) Bohr model, set to 2 electrons, instead of 1 electron for stability?. $\begingroup$ @Karl instructor went straight to drawing the Bohr model,. Aug 30, 2019 at 1:54 $\begingroup$ Well, Bohr is only step one. It seems you have progressed now, keep it up! $\endgroup$ - Karl. Aug 30, 2019 at 6:33. Add.
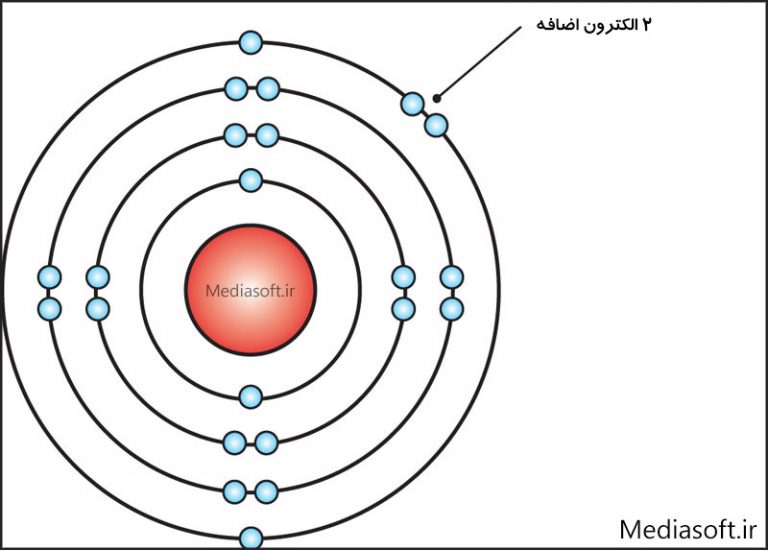
اتم چیست؟ سوالی در مورد فیزیک! مدیاسافت
In this video we'll look at the atomic structure and Bohr model for the Calcium atom (Ca). We'll use a Bohr diagram to visually represent where the electrons are around the nucleus.more.
Calcium Phosphate Nanoparticles AMERICAN ELEMENTS
Immediately before 1913, the Rutherford model conceived of an atom as consisting of a tiny positively charged heavy core, called a nucleus, surrounded by light, planetary negative electrons revolving in circular orbits of arbitrary radii. Britannica Quiz Matter and More Quiz How does Niels Bohr's atomic model work?

Calcium Bohr Model Project
The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits. Today, we know that the Bohr Model has some inaccuracies, but it's still used because of its simple approach to atomic theory.
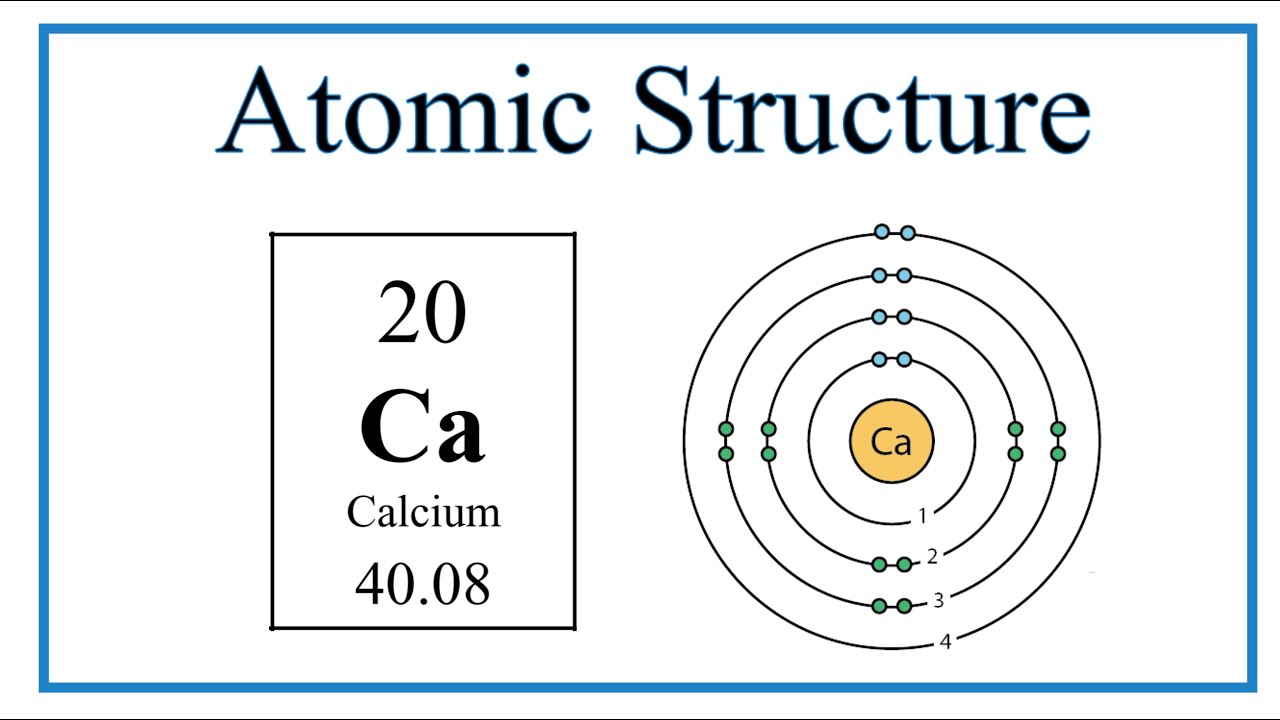
Calcium Shell Model
The Bohr model of calcium is a simplified representation of the atom's nuclear structure, named after Danish physicist Niels Bohr. It depicts the nucleus as a small, positively charged ball with electrons orbiting around it in circular paths at fixed distances.

Bohr Diagram Of Calcium
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.
Bohr Diagram Of Calcium
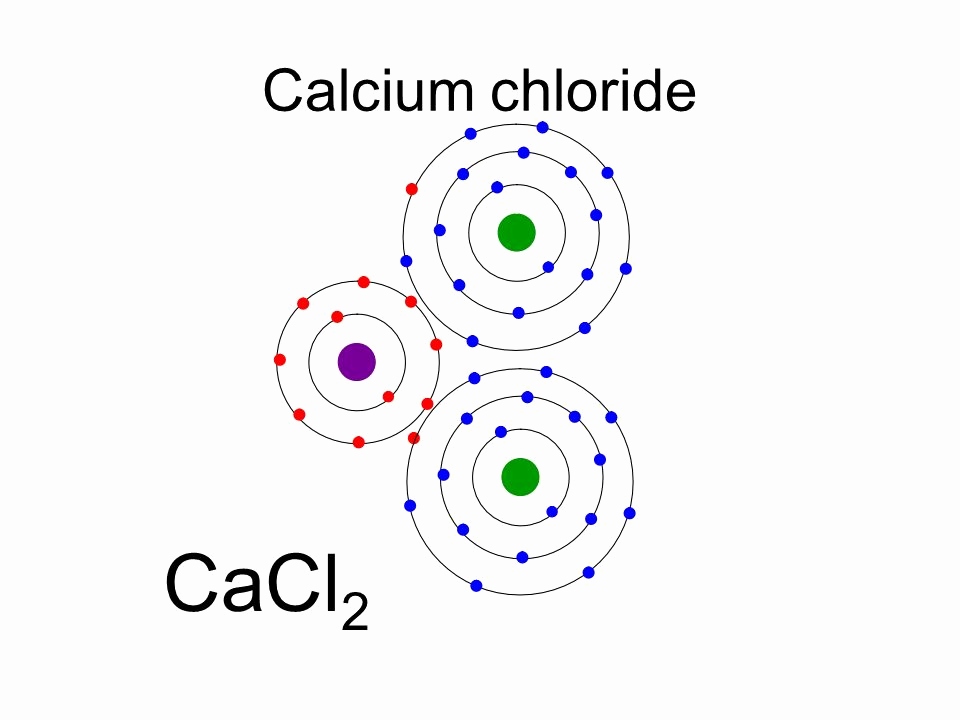
The elements that form bonds by donating electrons are called cation. Calcium donates two electrons of the last shell to form bonds and turns into a calcium ion (Ca 2+ ). That is, calcium is a cation element. Ca - 2e - → Ca 2+. The electron configuration of calcium ion (Ca 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6.

How to Build a Model of a Calcium Atom Articles MerchantCircle
The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory.
How can I draw electronic configuration of calcium in a shell nxwe70dd
An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Electrons are represented by dots or crosses and are positioned in energy levels, or 'shells', around the central nucleus. This is sometimes called the Bohr, or the 'solar system', model. Download this

Bohr Model Manganese Atom Electron Structure เวกเตอร์สต็อก (ปลอดค่า
Bohr model of calcium: (CC BY-SA 2.0 uk;Greg Robson): Answer b. Bohr model of sulfur: (CC BY-SA 2.0 uk; Greg Robson). Valence electrons are located in the highest energy level of an atom. When drawing a Bohr diagram, the valence electrons would be present in the outermost electronic level/shell (furthest away from the nucleus). An atom can have.

Calcium atom bohr model Royalty Free Vector Image
One of the weaknesses of Bohr's model was that he could not offer a reason why only certain energy levels or orbits were allowed. Figure 10.4.1 10.4. 1: The energy levels of the electrons can be viewed as rungs on a ladder. Note that the spacing between rungs gets smaller at higher energies (CC BY-NC; Ümit Kaya)

27 Bohr Diagram For Calcium Wiring Database 2020
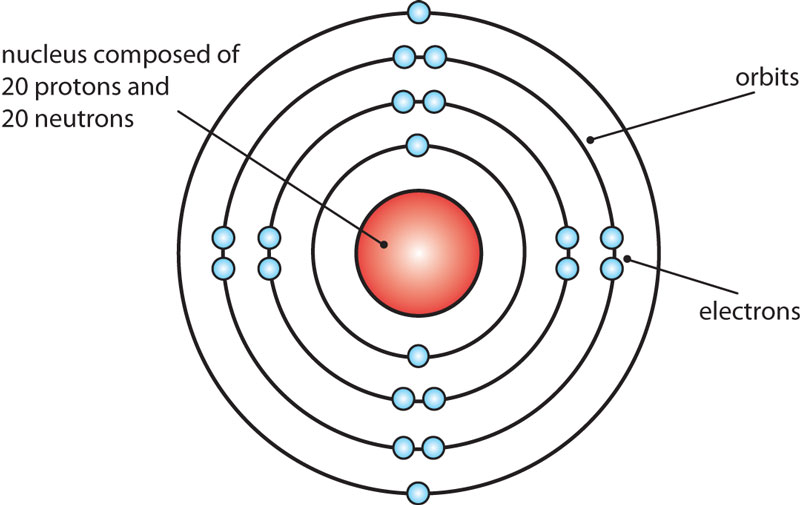
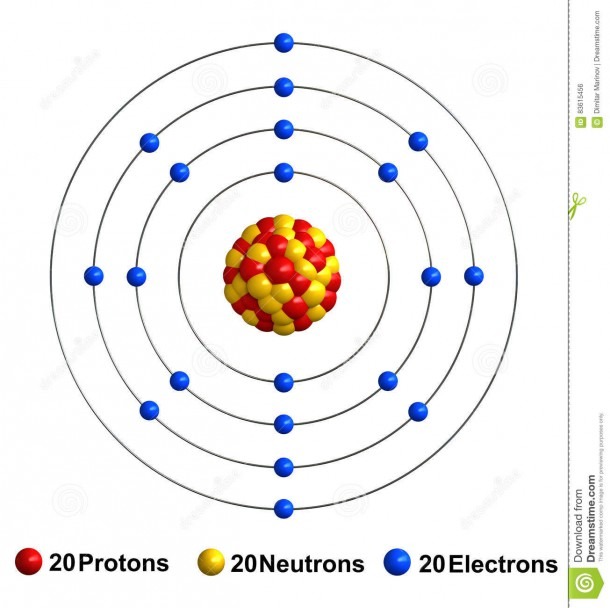
Name: Calcium Symbol: Ca Atomic Number: 20 Atomic Mass: 40.078 amu Melting Point: 839.0 °C (1112.15 K, 1542.2 °F) Boiling Point: 1484.0 °C (1757.15 K, 2703.2 °F) Number of Protons/Electrons: 20 Number of Neutrons: 20 Classification: Alkaline Earth Crystal Structure: Cubic Density @ 293 K: 1.55 g/cm 3 Color: Silvery Atomic Structure

Calcium Bohr Diagram
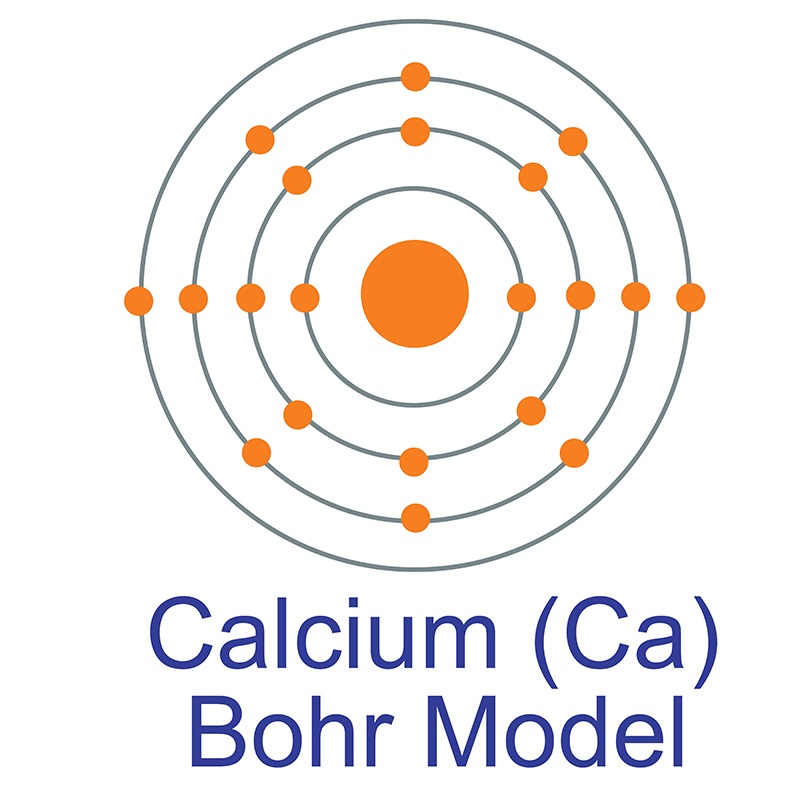
The Bohr Model of Calcium (Ca) has a nucleus that contains 20 neutrons and 20 protons. This nucleus is surrounded by four-electron shells named K-shell, L-shell, M-shell, and N-shell. The outermost shell in the Bohr diagram of Calcium contains only 2 electrons that also called valence electrons. Page Contents show
[DIAGRAM] Bohr Model Diagram For Calcium
Course: Class 9 Chemistry (India) > Unit 4. Lesson 1: Models of an atom. Discovery of the electron and nucleus. Rutherford's gold foil experiment. Drawback of the Rutherford model. Bohr's model of an atom. Atomic structure. Science >. Class 9 Chemistry (India) >.